Audit of medical devices, made easy
Audits that are 10 times faster, more secure documentation, and clearer decisions, thanks to AI-assisted pre-audits and automated compliance checks.
Awarded by the Hacking Health Camp
A full audit completed in just a few hours instead of several days.
Drastically reduce the risk of rejection from Stage 1
What if a virtual auditor could save you audit time and secure your compliance records?
Achieving and maintaining regulatory compliance for medical devices remains a major challenge: nearly one-third of applications fail at Stage 1, often for avoidable reasons. Manufacturers lose peace of mind, consultants spend days on repetitive reviews, and auditors waste valuable time on tedious checks.
Gordios is changing the game with its AI-powered virtual auditor. It thoroughly analyzes the QMS and technical file, detects discrepancies, classifies them by criticality, and offers concrete recommendations. It also manages additional manual reviews and generates a comprehensive report that includes traceability to standards and regulations.
Result: fewer rejections, more time saved, and secure audits for everyone — medical device manufacturers, QARA consultants, and notified bodies.


+10 years' experience
in medical devices
Key features
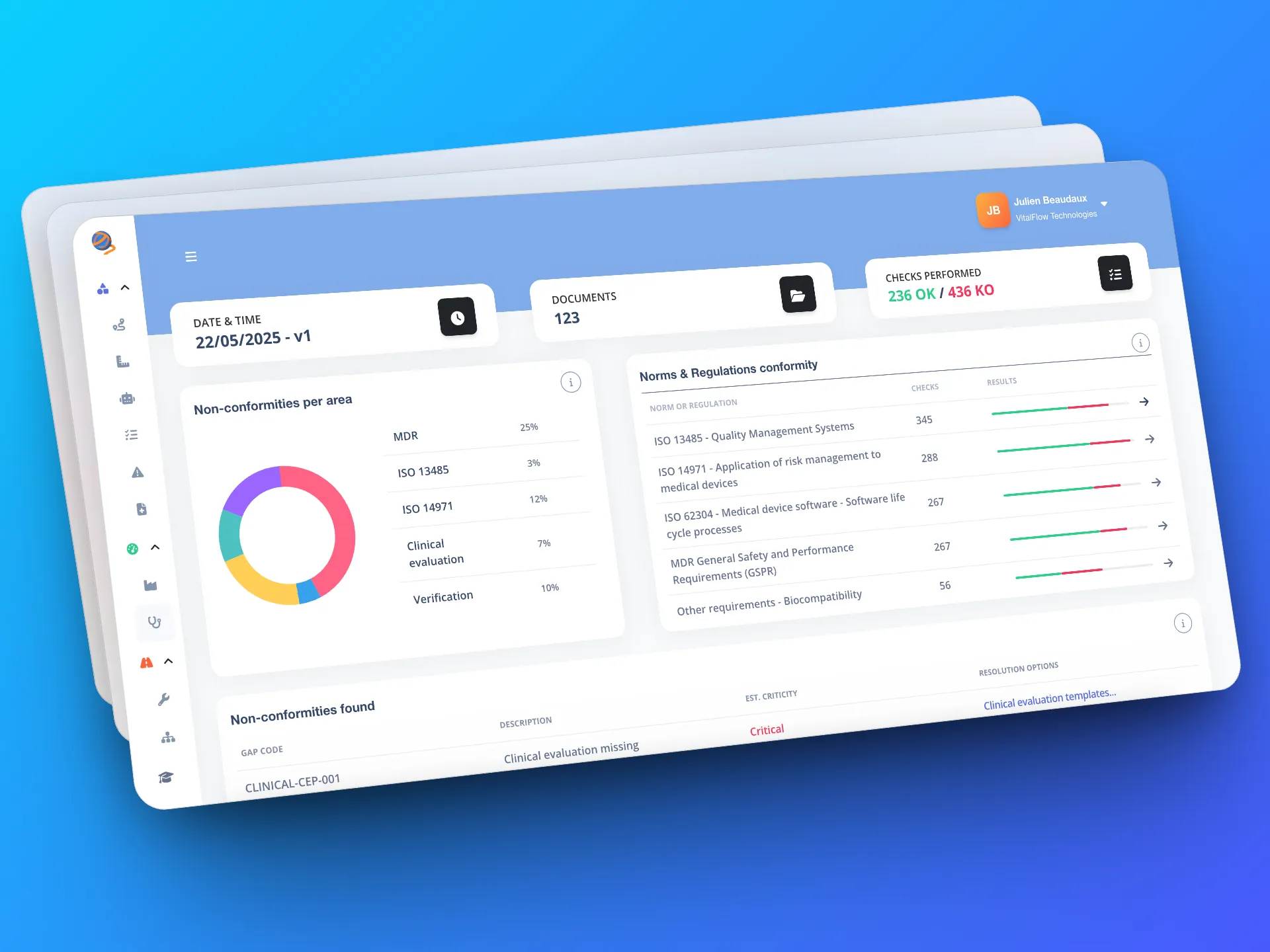
A comprehensive document review, in just a few minutes
Our AI reviews your QMS and technical file in just a few hours, whereas a manual analysis takes several days. Each document is checked against regulatory and standard requirements.
Result: a comprehensive, objective, and normative assessment of your compliance, with a clear prioritization of the gaps detected.
- Time savings: an audit in a few hours, compared to several days.
- In-depth analysis: several hundred analysis points systematically checked.
- Complete overview: EU MDR, ISO 13485, ISO 14971, and IEC 62304, ...
Clear, traceable, and actionable reports
Each audit generates a detailed report with figures, including an overall compliance rate, scores per section, discrepancies classified by criticality, etc. There is complete traceability, with each analysis linked to the corresponding standard or regulation.
This gives you a clear, documented, and immediately actionable overview to prepare your audits and demonstrate your compliance efforts.
- Detailed reports: scores, corrective actions, and gaps ranked by criticality.
- Full traceability: each analysis point linked to the corresponding rule.
- Operational clarity: a documented and immediately actionable overview for your audits and corrective actions.
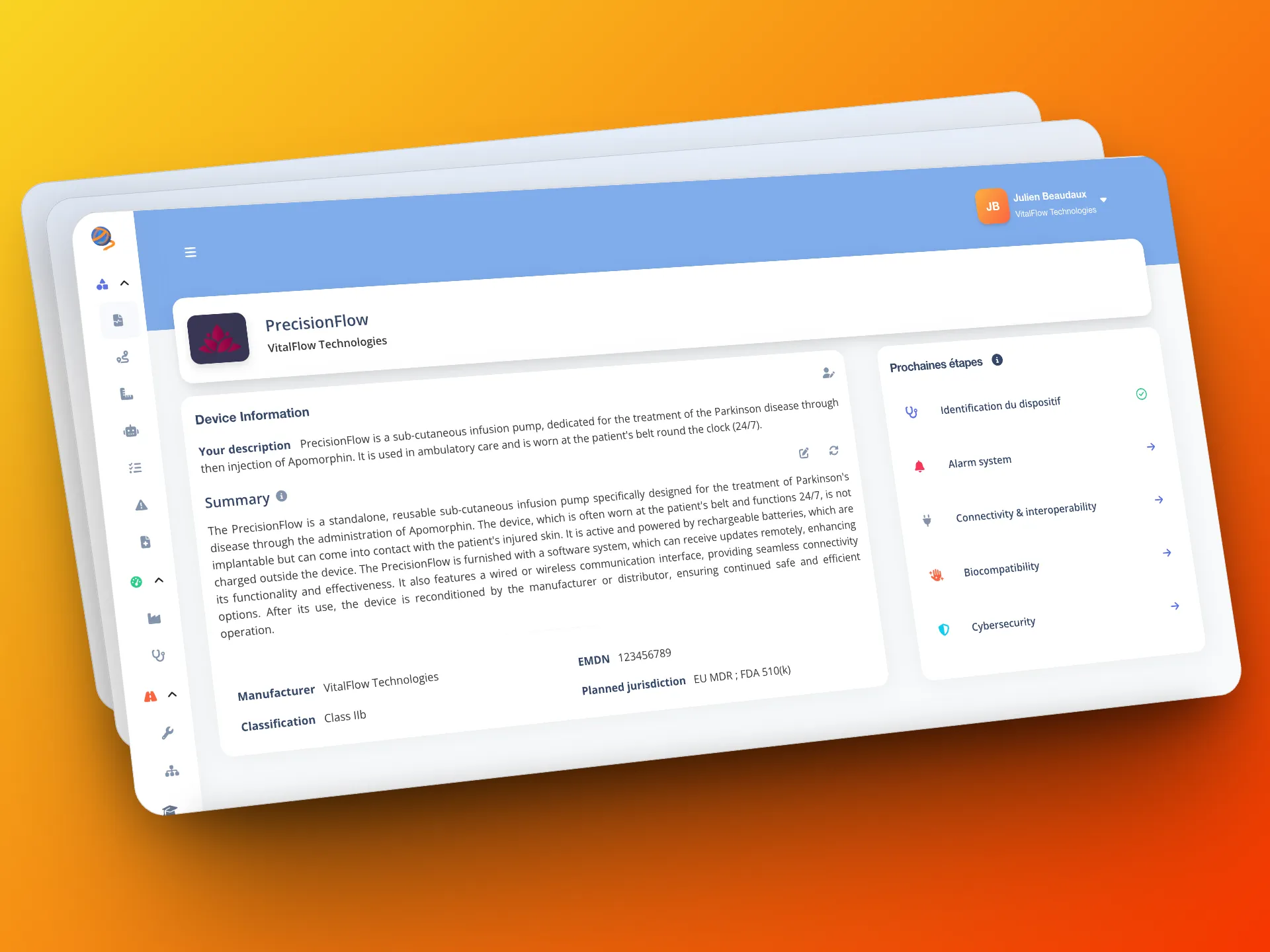
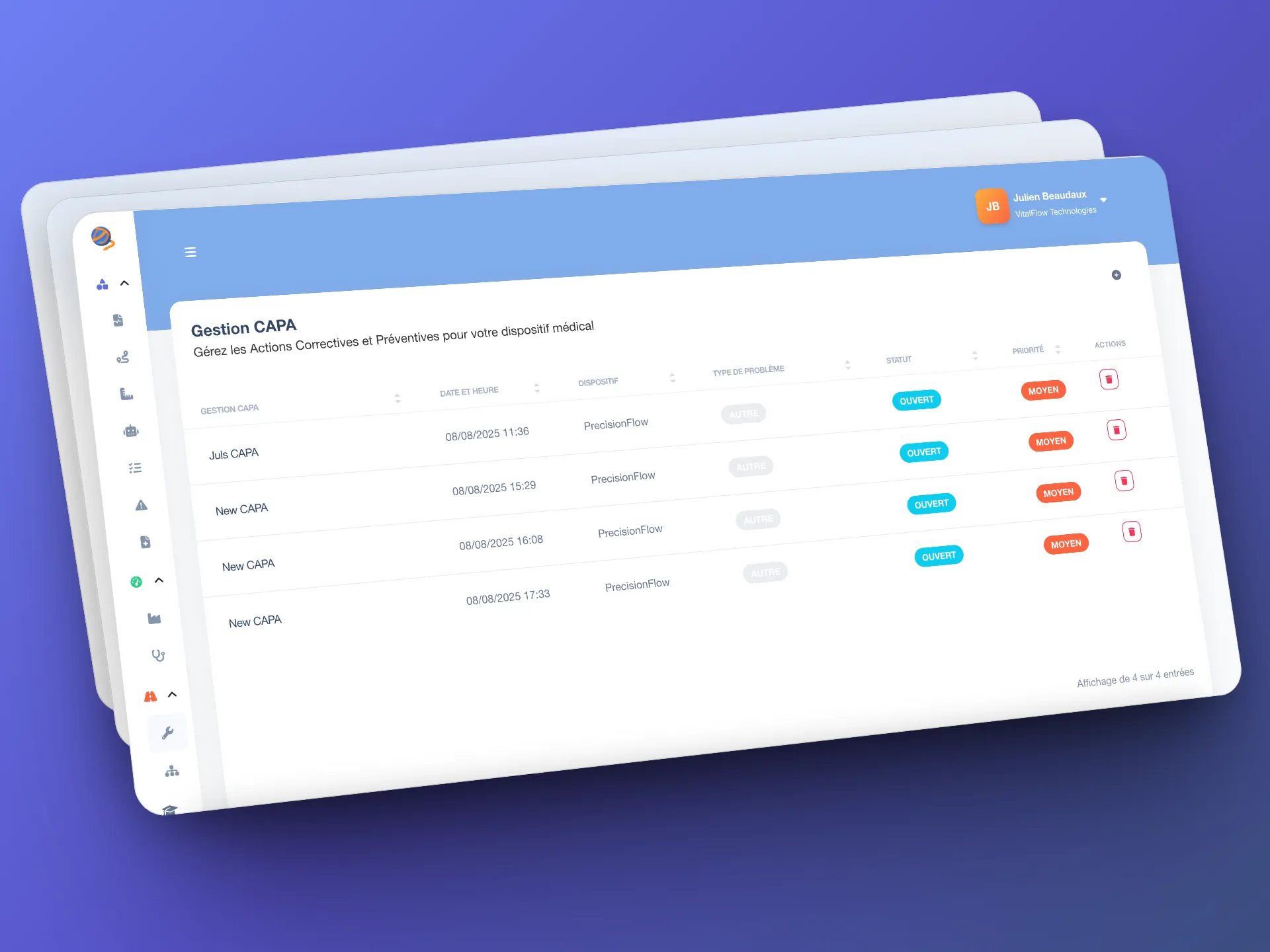
Automation enhanced by human expertise
Because human expertise remains essential, certain analyses are accompanied by manual validations, which you can entrust to your own QARA auditors/consultants or delegate to our Gordios experts. Gordios also provides customized checklists to cover points that require expert judgment and structure the rest of the audit.
This gives you the optimal balance between automation and expert validation.
- Customized checklists: a tool for structuring the rest of the audit.
- Expert supplement: certain analyses trigger validation by an expert.
- Flexibility of use: complete the audit with Gordios experts or your own.
Want to learn more?
Schedule a meeting with one of our specialists to best address your needs!
Book a demoThey trust us

Sarah Souheil Gebai
CEO TrembLess
My first impression of Gordios was shaped by the quality of the contact — not someone trying to sell me a service, but someone genuinely focused on guiding me through the specifications of my medical device. Before discovering Gordios, I had reached out to seven firms to estimate the cost of obtaining CE marking for my Class I device. The market price I was quoted ranged from €60,000 to €70,000. I assumed that was standard — until I found the solutions Gordios offered. Their pricing is highly competitive, and reaching out to them saved me valuable time. I highly recommend connecting with them early in your project.

Thomas Balverde
Regulatory Affairs Manager
I really appreciated the immediate relevance of Gordios' audit. Where I expected a simple document verification software, I discovered a true digital auditor capable of identifying discrepancies in my technical file in just a few minutes. Before, each revision of my QMS required several days of work, with a significant degree of uncertainty. I saved time, money, and above all, peace of mind. I cannot recommend Gordios highly enough before any audit!

Andrea Ballarin
CEO Hippocrate care
More than a solution, Gordios is a support and a partner in the development of your medical devices. We saved a lot of time and money by integrating Gordios right from the start of our development. A regulatory overview, but above all, a 360° view with experts, all in the blink of an eye. If you're a healthcare start-up, you should get in touch with the team!

Dr Céline Besnier
CEO SportSantéClic
Gordios has been a game-changer for me in setting-up our quality system. They provide an easy approach for a non-tech person like me and clearly reduced my time-to-market, while making my solution conform with all contract requirements.



